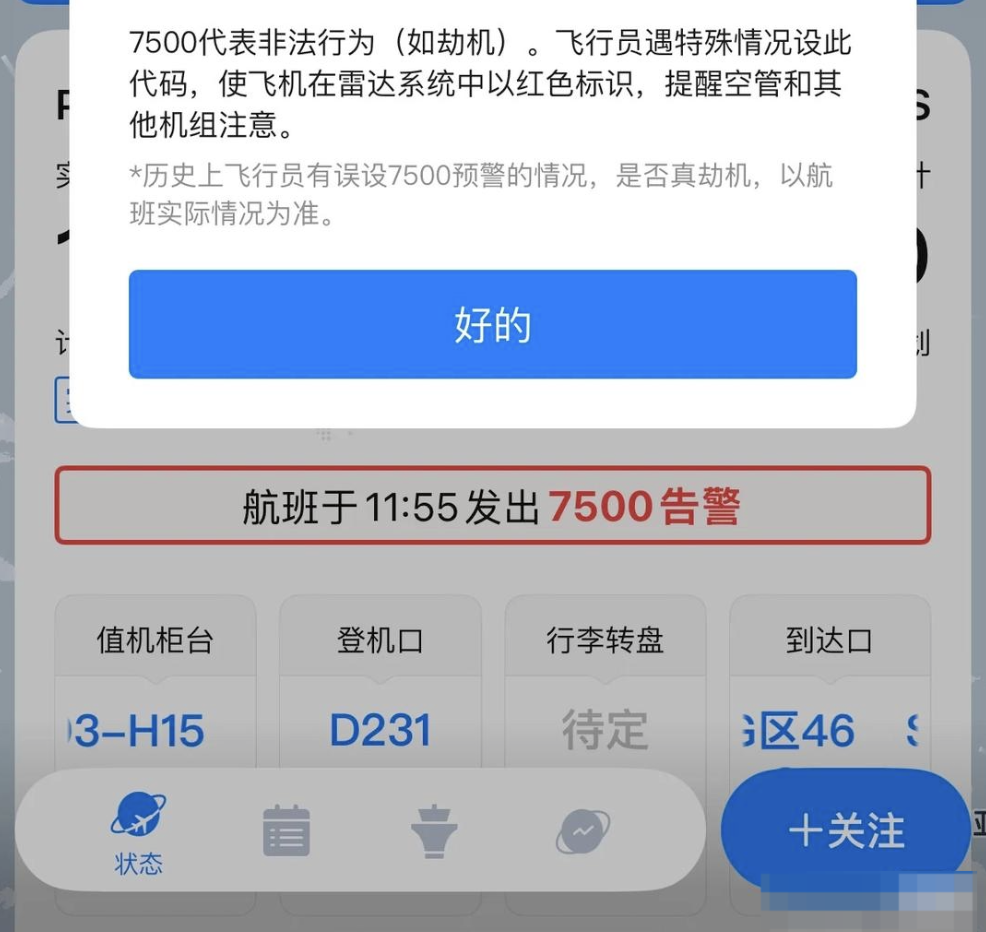
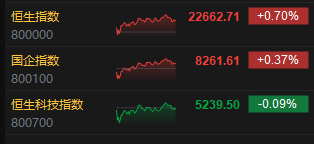
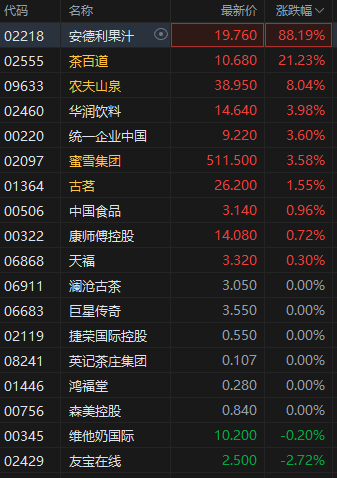
港聞更多
中國新聞更多
焦點娛聞
體育報導

今日財經

515投資者保護日|金鷹基金走進成都信息工程大學:為高校師生呈現了一場願意聽、聽得懂、用得上的線下活動
新浪財經“2025年度投教優秀案例評選”活動已於3月21日正式啟動。金鷹基金走進成都信息工程大學線下…
BEAUTY

卡地亞攜手HalleBerry、Rihanna、MileyCyrus、BadBunny、KarlieKloss、AnnaSawai、AimeeLouWood等一眾巨星於2025年MetGala紐約大都會博物館慈善舞會的創意舞台上以標志性美學風格與精湛工藝
#鐘表與奇跡2025#【#metgala紅毯#】卡地亞攜手Halle Berry、Rihanna、M…