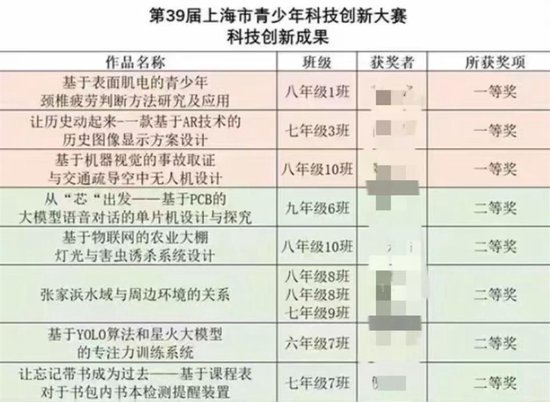
港聞更多
中國新聞更多
焦點娛聞

BTSPOP-UP:MONOCHROME,2024.04.26.-05.1211AM-8PM期待期待~
【Twi】BTS POP-UP : MONOCHROME,2024.04.26. – 0…
體育報導
今日財經
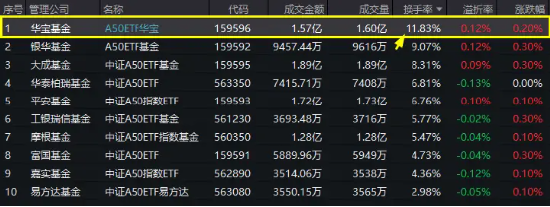
正式納入兩融標的!A50ETF華寶(159596)交投尤為活躍,換手率飆至11.83%,高居同類第一!場內放量溢價
4月17日,A股顯著回暖,核心龍頭股大面積飄紅,中證A50指數盤中成功翻紅,成份股東方雨虹、紫光股份…
BEAUTY

有錢人都這樣穿?!Netflix《淚之女王》金智媛的五種「財閥千金穿搭套路」解析~墊肩西外不可少!
超熱播的《淚之女王》你追了嗎!其中金智媛在裏頭所飾演的財閥千金「洪海仁」可謂是引人注目的焦點~每次亮…
生活消閒

起床不能馬上喝咖啡!久了恐發胖?營養師、英國飲食專家教你如何用五步驟開啟精神飽滿的一天♡
早上的心情和精神狀態往往會影響整天的表現和情緒~所以建立一個正面、健康的早晨習慣至關重要!無論你是準…